Efficient production of lactic acid from sugars over Sn-Beta zeolite in water: catalytic performance and mechanistic insights†
Abstract
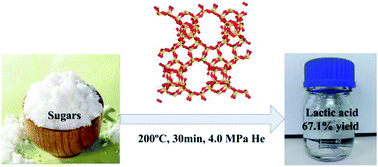
Template-free synthesis of Sn-Beta zeolite was realized by oil-heated crystallization within 36 h. The Sn-Beta zeolite with a high surface area of 623 m2 g−1 can efficiently catalyze the one-pot hydrothermal conversion of various sugars to lactic acid. Under the reaction conditions of 200 °C with a helium pressure of 4.0 MPa, the conversion of all sugars exceeds 98% within 30 min, and the yield of lactic acid reaches as high as 67.1%. For glucose conversion to lactic acid, further experiments demonstrate the essential role of the Lewis acid site at the different stages of the reaction, which is attributed to the isolated skeleton Sn in the zeolite, consistent with our and previous computational studies on this reaction. Our calculations further reveal the important role of the Brønsted acid site of moderate strength, such as lactic acid itself, in the conversion of glyceraldehyde to pyruvaldehyde, an important step in the formation of lactic acid from glucose, resulting in self-catalysis.
- This article is part of the themed collection: 2019 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...