Macrocyclisation of small peptides enabled by oxetane incorporation†
Abstract
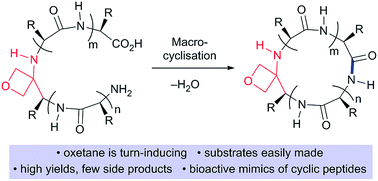
Cyclic peptides are an important source of new drugs but are challenging to produce synthetically. We show that head-to-tail peptide macrocyclisations are greatly improved, as measured by isolated yields, reaction rates and product distribution, by substitution of one of the backbone amide C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds with an oxetane ring. The cyclisation precursors are easily made by standard solution- or solid-phase peptide synthesis techniques. Macrocyclisations across a range of challenging ring sizes (tetra-, penta- and hexapeptides) are enabled by incorporation of this turn-inducing element. Oxetane incorporation is shown to be superior to other established amino acid modifications such as N-methylation. The positional dependence of the modification on cyclisation efficiency is mapped using a cyclic peptide of sequence LAGAY. We provide the first direct experimental evidence that oxetane modification induces a turn in linear peptide backbones, through the observation of dNN (i, i + 2) and dαN (i, i + 2) NOEs, which offers an explanation for these improvements. For cyclic peptide, cLAGAY, a combination of NMR derived distance restraints and molecular dynamics simulations are used to show that this modification alters the backbone conformation in proximity to the oxetane, with the flexibility of the ring reduced and a new intramolecular H-bond established. Finally, we incorporated an oxetane into a cyclic pentapeptide inhibitor of Aminopeptidase N, a transmembrane metalloprotease overexpressed on the surface of cancer cells. The inhibitor, cCNGRC, displayed similar IC50 values in the presence or absence of an oxetane at the glycine residue, indicating that bioactivity is fully retained upon amide C
O bonds with an oxetane ring. The cyclisation precursors are easily made by standard solution- or solid-phase peptide synthesis techniques. Macrocyclisations across a range of challenging ring sizes (tetra-, penta- and hexapeptides) are enabled by incorporation of this turn-inducing element. Oxetane incorporation is shown to be superior to other established amino acid modifications such as N-methylation. The positional dependence of the modification on cyclisation efficiency is mapped using a cyclic peptide of sequence LAGAY. We provide the first direct experimental evidence that oxetane modification induces a turn in linear peptide backbones, through the observation of dNN (i, i + 2) and dαN (i, i + 2) NOEs, which offers an explanation for these improvements. For cyclic peptide, cLAGAY, a combination of NMR derived distance restraints and molecular dynamics simulations are used to show that this modification alters the backbone conformation in proximity to the oxetane, with the flexibility of the ring reduced and a new intramolecular H-bond established. Finally, we incorporated an oxetane into a cyclic pentapeptide inhibitor of Aminopeptidase N, a transmembrane metalloprotease overexpressed on the surface of cancer cells. The inhibitor, cCNGRC, displayed similar IC50 values in the presence or absence of an oxetane at the glycine residue, indicating that bioactivity is fully retained upon amide C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond replacement.
O bond replacement.
- This article is part of the themed collection: Editor’s choice – Andrei Yudin


 Please wait while we load your content...
Please wait while we load your content...