A nonaqueous potassium-ion hybrid capacitor enabled by two-dimensional diffusion pathways of dipotassium terephthalate†
Abstract
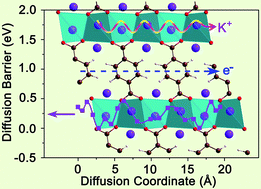
Nonaqueous potassium-ion hybrid capacitors (KIHCs) are faced with limited redox reaction kinetics of electrodes for accommodation of large-sized K+. Here, dipotassium terephthalate (K2TP) is applied as an organic negative electrode to provide comparable reaction kinetics with a non-faradaic activated carbon (AC) positive electrode to boost the electrochemical performance of KIHCs. It is revealed that the large exchange current density and fast two-dimensional (2D) diffusion pathways of K+ in K2TP determined by density functional theory (DFT) calculations ensure its fast redox reaction and transport kinetics. The as-constructed KIHC presents both high energy and power densities of 101 W h kg−1 and 2160 W kg−1 based on the mass of the two electrodes (41.5 W h kg−1 and 885.2 W kg−1 based on the mass of the two electrodes and electrolyte), respectively, and a superior capacity retention of 97.7% after 500 cycles. The excellent electrochemical performance is attributed to the fast kinetics, good structural flexibility, and small volume change (9.4%) of K2TP upon K+ insertion/extraction, and its good compatibility with the AC positive electrode in 1,2-dimethoxyethane (DME)-based electrolyte. This will promote application of organic materials in hybrid capacitors and the development of KIHCs.
- This article is part of the themed collection: Celebrating 100 Years of Chemistry at Nankai University


 Please wait while we load your content...
Please wait while we load your content...