Effect of salt on the formation of 5-hydroxymethylfurfural from ketohexoses under aqueous conditions
Abstract
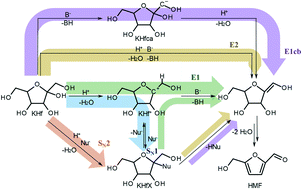
5-Hydroxymethylfurfural (HMF) is a valuable platform chemical that can be produced by an acid-catalysed triple dehydration of hexoses, in particular ketoses. The mechanism of HMF formation is not fully understood, i.a. it has been hypothesised that anions could be involved in the reaction, provided that they are present. In this work, the effect of different monovalent, weakly nucleophilic anions on the HMF formation from fructose and tagatose is studied in order to elucidate their possible role in the mechanism. All anions investigated cause an acceleration of the initial HMF formation compared with the salt-free sample, i.e. there is a reaction order with respect to the anion concentration between 0.12 and 0.68. Merely higher concentrations of nitrate inhibited the HMF formation. Furthermore, by studying the insertion of 18O from labelled water into fructose using LC-MS, a hydroxyl group exchange could be identified giving rise to the assumption that substitution reactions play an important role.


 Please wait while we load your content...
Please wait while we load your content...