Fluorine ion induced phase evolution of tin-based perovskite thin films: structure and properties†
Abstract
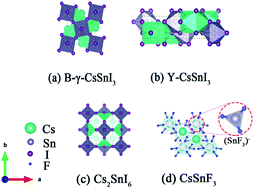
To study the effect of fluorine ions on the phase transformation of a tin-based perovskite, CsSnI3−x(F)x films were deposited by using thermal vacuum evaporation from a mixed powder of SnI2, SnF2 and CsI, followed by rapid vacuum annealing. The color evolution, structure, and properties of CsSnI3−xFx films aged in air were observed and analyzed. The results showed that the colors of the films changed from black to yellow, and finally presented as black again over time; the unstable B-γ-CsSnI3−xFx phase transformed into the Y-CsSnI3−xFx phase, which is then recombined into the Cs2SnI6−xFx phase with the generation of SnO2 in air. Fluorine dopant inhibited the oxidation process. The postponement of the phase transformation is due to the stronger bonds between F and Sn than that between I and Sn. The color changing process of the CsSnI3−xFx films slowed that the hole concentrations increased and the resistivities decreased with the increase of the F dopant ratio. With the addition of SnF2, light harvesting within the visible light region was significantly enhanced. Comparison of the optical and electrical properties of the fresh annealed CsSnI3−xFx films showed that the band gaps of the aged films widened, the hole concentrations kept the same order, the hole mobilities reduced and therefore, the resistivities increased. The double layer Cs2SnI6−xFx phase also showed ‘p’ type semi-conductor properties, which might be due to the incomplete transition of Sn2+ to Sn4+, i.e. Sn2+ provides holes as the acceptor.


 Please wait while we load your content...
Please wait while we load your content...