Photolysis of carboxymethylflavin in aqueous and organic solvent: a kinetic study†
Abstract
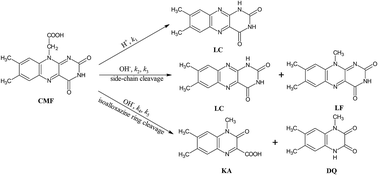
This is the first study on the photolysis of carboxymethylflavin (CMF), an intermediate in the photolysis of riboflavin (RF). CMF is photodegraded by removal of side-chain to lumichrome (LC) in acid solution and to LC and lumiflavin (LF) in alkaline solution. It also undergoes alkaline hydrolysis to 1,2-dihydro-1-methyl-2-keto-3-quinoxaline carboxylic acid (KA) and 1,2,3,4-tetrahydro-1-methyl-2,3-dioxoquinoxaline (DQ) by cleavage of isoalloxazine ring. CMF degrades to LC in organic solvents. The formation of LC in acid solution and organic solvents takes place by second-order reaction and those of LC, LF, KA and DQ in alkaline solution by first-order reactions. The values of second-order rate constants for the photolysis of CMF at pH 2.0 to 7.0 are in the range of 1.13 to 2.45 M−1 s−1 and those of first-order rate constants (kobs) at pH 8.0–12.0 from 1.53 to 4.18 × 10−4 s−1 and for the formation of photoproducts from 0.37 to 16.6 × 10−5 s−1. The photolysis of CMF is enhanced, with pH, in the alkaline region since the excited state is sensitive to alkaline hydrolysis. The photolysis and fluorescence quantum yields of CMF in aqueous and organic solvents have been reported. CMF and photoproducts have been assayed spectrofluorimetrically. The mode of CMF photolysis is discussed.


 Please wait while we load your content...
Please wait while we load your content...