Trichalcogenasumanenes containing various chalcogen atoms: synthesis, structure, properties, and chemical reactivity†
Abstract
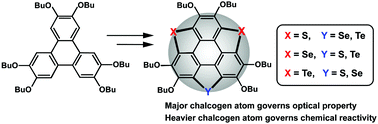
Trichalcogenasumanenes (1–3) are rich in chemistry, as they can be regio-selectively functionalized. Chalcogen atoms (S, Se, and Te) play a pivotal role in determining molecular geometry, optical properties, and reactivity. Herein, new trichalcogenasumanenes (4–9) are synthesized, all with the benzylic carbons on sumanene replaced by two kinds of chalcogen atom. Crystallographic studies show that the molecular geometry varies from bowl-shaped (4 and 6) to planar (9) as the size of the majority chalcogen atom increases from S (4) to Se (6) to Te (9). The optical properties of sulfur-rich (4–5), selenium-rich (6–7), and tellurium-rich (8–9) examples are respectively similar to those of trithia- (1), triselena- (2), and tritellura-sumanenes (3). The chemical activities of 4–7 are evaluated via reactions with Oxone, H2O2, and Br2. It is found that their reactivities are dominated by the heavier chalcogen atom rather than the one in the majority.


 Please wait while we load your content...
Please wait while we load your content...