Syntheses of 2-(2,2,2-trifluoroethylidene)/(2,2-difluoroethyl)-1,3-dicarbonyl compounds and their fungicidal activities†
Abstract
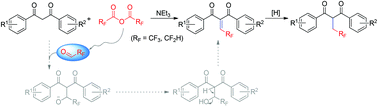
A Knoevenagel-type reaction of 1,3-diketone substrates with trifluoroacetic anhydride is described. The electrophilic species of trifluoroacetaldehyde is easily generated in situ by the reaction of trifluoroacetic anhydride/difluoroacetic anhydride with triethylamine. By this efficient and simple approach, a library of 2-(2,2,2-trifluoroethylidene)-1,3-dicarbonyl compounds that contain a wide range of functional groups were synthesized in good yields. Moreover, the reaction with difluoroacetic anhydride affords the corresponding (2,2-difluoroethyl)-1,3-dicarbonyls. Some of these compounds exhibited promising fungicidal activities.


 Please wait while we load your content...
Please wait while we load your content...