Hydrogels with multiple clickable anchor points: synthesis and characterization of poly(furfuryl glycidyl ether)-block-poly(ethylene glycol) macromonomers†
Abstract
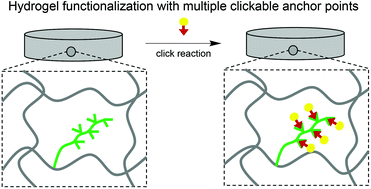
Functional polyacrylamide hydrogels containing multiple furfuryl anchor points for Diels–Alder reactions were prepared employing new macromonomers. For this purpose, α-diphenylmethyl-ω-4-vinyl benzyl-poly(furfuryl glycidyl ether)-block-poly(ethylene glycol) (PFGEp-b-PEGq) macromonomers were synthesized via anionic ring opening polymerization in four different block lengths: PFGE10-b-PEG25, PFGE8-b-PEG79, PFGE18-b-PEG66 and PFGE13-b-PEG111. These furan containing building blocks were characterized by 1H NMR, 13C NMR and FT-IR spectroscopy, size exclusion chromatography and MALDI TOF mass spectrometry and revealed low molar mass dispersities between 1.05 and 1.09 and number average molar masses between 2330 g mol−1 and 6660 g mol−1. The block lengths of the macromonomers were adjustable via the monomer-to-initiator ratio and high end group functionalization degrees between 72% and 98% were achieved. The macromonomers were characterized by DSC and TGA regarding their thermal properties (−43 °C ≤ Tg ≤ −32°, 32 °C ≤ Tm ≤ 45 °C, 369 °C ≤ Td ≤ 381 °C) and by tensiometry regarding their surface activity, revealing a critical micelle concentration around 0.3 mg mL−1 in aqueous solution. The furfuryl groups of the PFGEp-b-PEGq macromonomers were reactive in Diels–Alder reactions in solution as well after their integration into polyacrylamide hydrogels by radical copolymerization, as shown by fluorescence labeling employing a maleimide-functionalized dye. Thus, a new synthetic route is opened up to access functional hydrogels providing clickable anchor points in high density for conjugation of maleimide-functional substrates.


 Please wait while we load your content...
Please wait while we load your content...