Polymerization of phenylacetylenes by binuclear rhodium catalysts with different para-binucleating phenoxyiminato linkages†
Abstract
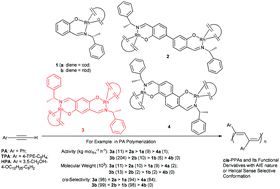
In contrast to the extensive studies on the cooperative effects of bimetallic olefin polymerization catalysts, less attention has been paid to binuclear rhodium (Rh) catalysts for alkyne polymerization and almost no binuclear Rh catalyst exhibits cooperative effects in phenylacetylene (PA) polymerization to date. Two series of binuclear Rh catalysts with different para-binucleating phenoxyiminato linkages of general formula [(diene)Rh]2(phenoxyiminato) (where diene = cod (a) or nbd (b), phenoxyiminato linkage = 4,4′-bisalicylaldiminato (2), 2,5-phenyloxydiiminato (3), and 2,6-naphthoxydiiminato (4)) have been synthesized in order to investigate their bimetallic cooperative effects in the polymerization of PA and its functional derivatives (such as hydroxyl containing PA (HPA) or tetraphenylethylene containing PA (TPA)). The polymerization results demonstrate that the catalytic characteristics of these binuclear Rh catalysts strongly depend on the catalyst structures modified by chelating ligands. Compared to the corresponding mononuclear salicylaldiminato Rh diene complexes (1(a–b)) in PA polymerization under identical conditions, binuclear 2,5-phenyloxydiiminato ligated Rh nbd catalyst 3b displays cooperative effects in terms of enhanced catalytic activity, cis-selectivity, and polymer molecular weight. Moreover, such cooperative effects of 3b could also be observed in the polymerization of functional phenylacetylenes such as 3,5-CH2OH-4-OC12H25 substituted phenylacetylene (HPA) or tetraphenylethylene (TPE) containing phenylacetylene (TPA), affording high molecular weight functional cis-PPAs with a helical sense selective conformation or AIE nature.


 Please wait while we load your content...
Please wait while we load your content...