The functionalization of poly(ε-caprolactone) as a versatile platform using ε-(α-phenylseleno) caprolactone as a monomer†
Abstract
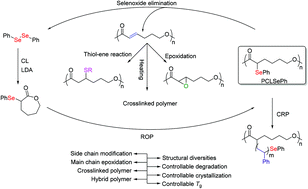
This paper describes a novel ε-caprolactone monomer modified by a phenylseleno group at the α-position of the carbonyl. Poly(α-phenylseleno caprolactone) (PCLSePh) with a well-controlled molecular weight and narrow molecular weight distribution (Đ < 1.20) was prepared through ring-opening polymerization. Hybrid polymers with properties superior to those of polyester materials were also prepared though selenide-mediated radical polymerizations under UV light irradiation. Subsequently, a carbon–carbon double bond was introduced into the main chain of the polymer by the oxidative elimination of the phenylseleno groups with high efficiency. Various side groups were further introduced into the polymer chain through a nucleophilic thiol–ene reaction. This strategy enables the synthesis of PCL with functional groups that allow the multifunctional integration of polyester materials. Furthermore, this monomer can be copolymerized with ε-caprolactone to form both random and block copolymers. The glass transition temperature, crystallinity and degradation rate of the copolymers can be adjusted both by oxidation and tuning the composition of the copolymer.


 Please wait while we load your content...
Please wait while we load your content...