Preparation and in situ chain-end-functionalization of branched ethylene oligomers by monosubstituted α-diimine nickel catalysts†
Abstract
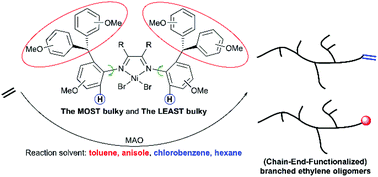
By utilizing an efficient strategy of the combination of the most bulky and the least bulky substituents in the ortho-N-aryl groups of α-diimine ligands, a new class of unsymmetrically monosubstituted (H/CHAr2) α-diimine nickel complexes Ni-1–5 and palladium complexes Pd-1–5 with trans- and cis-forms was synthesized and comprehensively identified by NMR spectroscopy, single crystal X-ray diffraction, mass spectroscopy, and elemental analysis. In the presence of excess MAO, all these nickel complexes Ni-1–5 were thermally stable (−10–70 °C) and exhibited extremely high activities of 106–107 g mol−1 h−1 for ethylene polymerization. By readily varying the reaction temperature, highly linear polyethylenes (<2/1000C, Tm = 132–140 °C) with moderate molecular weights (Mn: 104 g mol−1) and (highly) branched ethylene oligomers (up to 73/1000C) with a Mn of 103 g mol−1 could be produced with broad bimodal molecular weight distributions, respectively. More remarkably, chain-end-functionalization of these generated oligoethylenes could be achieved in situ by the Friedel–Crafts addition to the aromatic compounds of the reaction solvent (such as toluene and anisole) and could be also tuned from 0% to 100% conversion with temperature, solvent, and catalyst. As a comparison, under the activation of NaBArF, all these palladium complexes Pd-1–5 displayed good activity (104 g mol−1 h−1) for ethylene polymerization to yield low molecular weight branched ethylene oligomers (52–70/1000C) and were also active for the copolymerization of ethylene and methyl acrylate (MA) to generate ethylene/MA co-oligomers with an ester functional group at the end of branches (0.87–3.65 mol%).


 Please wait while we load your content...
Please wait while we load your content...