Stearoyl-appended pendant amino acid-based hyperbranched polymers for selective gelation of oil from oil/water mixtures†
Abstract
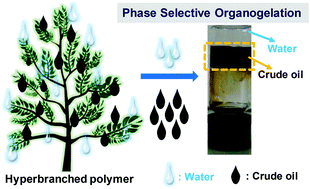
Herein, we demonstrate the architecture-directed self-assembly of a hyperbranched polymer-based organogelator, resulting in the phase-selective organogelation of crude oil from an oil/water biphasic mixture. Hyperbranched polymeric organogelators comprising stearic acid-appended pendant L-amino acid-based poly(methacrylate)s were synthesized via the combination of self-condensing vinyl polymerization (SCVP) and reversible addition–fragmentation chain transfer (RAFT) polymerization. Five hyperbranched polymers based on five different L-amino acids such as alanine (Ala), valine (Val), leucine (Leu), isoleucine (Ile) and phenylalanine (Phe) were synthesized to study the effect of the pendant –R groups of amino acids on the phase-selective organogelation of the polymers in various organic solvents and fuel oils. Among the five hyperbranched polymers, only the Ala- and Val-containing polymers selectively congealed aliphatic organic hydrocarbons and fuel oils, including diesel, kerosene and crude oil. The morphology of the self-assembled organogels was characterized by field-emission scanning electron microscopy (FE-SEM) and transmission electron microscopy (TEM), and their mechanical strength was determined by rheological measurements. FT-IR spectroscopy revealed hydrogen bonding-directed self-assembly in the polymer matrix, which could be the main reason for the formation of the fibrous network and thus gelation in selective organic hydrocarbons immobilizing oil or organic solvents in the interstices of the matrix.


 Please wait while we load your content...
Please wait while we load your content...