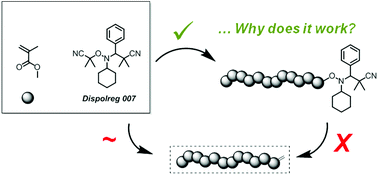
Why can Dispolreg 007 control the nitroxide mediated polymerization of methacrylates?†
Abstract
The alkoxyamine 3-(((2-cyanopropan-2-yl)oxy)(cyclohexyl)amino)-2,2-dimethyl-3-phenylpropanenitrile (Dispolreg 007) has recently emerged as a robust regulator for the controlled (co)polymerization of methacrylates and styrene by nitroxide mediated polymerization. However, whilst the successful use of this alkoxyamine in a number of systems has now been demonstrated, the fundamental reason behind its ability to overcome the long-standing issues that normally arise in the nitroxide mediated polymerization of methacrylates has not been conclusively determined. Herein, a combination of quantum chemical calculations, mathematical modelling and experimental data is utilized to compare the behaviour of Dispolreg 007 with alternative nitroxides such as SG1 and DPAIO. The chemical structure of Dispolreg 007 leads to a nitroxide which has a higher rate of combination and a lower rate of β-H transfer with the propagating radical compared to SG1. This is not the case for DPAIO which shows relatively similar rate constants. The comparison between modelled and experimental data allowed for a better understanding of the system, with small amounts of macromonomers being produced mainly by β-H transfer at high monomer conversions (>80%). Finally, the estimation of the rate constants was key to optimize the polymerization conditions to achieve good control and high livingness at high monomer conversions (90%).


 Please wait while we load your content...
Please wait while we load your content...