Synthesis, spectroscopy and QM/MM simulations of a biomimetic ultrafast light-driven molecular motor†‡
Abstract
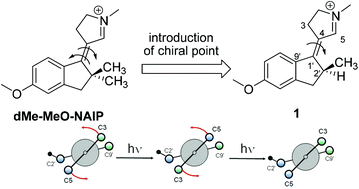
A molecular motor potentially performing a continuous unidirectional rotation is studied by a multidisciplinary approach including organic synthesis, transient spectroscopy and excited state trajectory calculations. A stereogenic center was introduced in the N-alkylated indanylidene–pyrroline Schiff base framework of a previously investigated light-driven molecular switch in order to achieve the unidirectional C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C rotary motion typical of Feringa's motor. Here we report that the specific substitution pattern of the designed chiral molecule must critically determine the unidirectional efficiency of the light-induced rotary motion. More specifically, we find that a stereogenic center containing a methyl group and a hydrogen atom as substituents does not create a differential steric effect large enough to fully direct the motion in either the clockwise or counterclockwise direction especially along the E → Z coordinate. However, due to the documented ultrafast character and electronic circular dichroism activity of the investigated system, we find that it provides the basis for development of a novel generation of rotary motors with a biomimetic framework and operating on a picosecond time scale.
C rotary motion typical of Feringa's motor. Here we report that the specific substitution pattern of the designed chiral molecule must critically determine the unidirectional efficiency of the light-induced rotary motion. More specifically, we find that a stereogenic center containing a methyl group and a hydrogen atom as substituents does not create a differential steric effect large enough to fully direct the motion in either the clockwise or counterclockwise direction especially along the E → Z coordinate. However, due to the documented ultrafast character and electronic circular dichroism activity of the investigated system, we find that it provides the basis for development of a novel generation of rotary motors with a biomimetic framework and operating on a picosecond time scale.
- This article is part of the themed collection: In memory of Professor Ugo Mazzucato (1929–2017)


 Please wait while we load your content...
Please wait while we load your content...