Solvent- and catalyst-free transamidations of unprotected glycosyl carboxamides†
Abstract
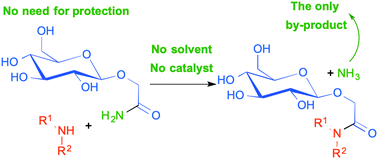
The transamidation reactions of unprotected mono- and disaccharidic carboxamides with various primary and secondary arylic, heterocyclic or aliphatic amines are described. This new method is green and atom efficient and gives good to high yields. Notably, the conditions do not require either a solvent or a catalyst and give ammonia as a single by-product. The described coupling reaction is compatible with a variety of functional groups and was used in the synthesis of various glycosidic derivatives and biologically relevant glycolipids. A plausible reaction mechanism involving an intermolecular H-bond activation of the starting carboxamides is proposed.
- This article is part of the themed collections: Chemical Biology in OBC and Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...