Synthesis of fluorenyl alcohols via cooperative palladium/norbornene catalysis†
Abstract
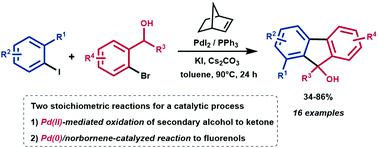
Herein, we report a novel catalytic synthesis of substituted 9H-fluoren-9-ols starting from aryl iodides and secondary ortho-bromobenzyl alcohols in the presence of palladium/norbornene as a catalytic system. The present protocol exhibits high functional group tolerance, mild reaction conditions and moderate to good yields. This transformation is based on two sequential pathways: (i) Pd(II)-mediated oxidation of the secondary alcohol to the corresponding ketone and (ii) Pd(0)/norbornene-catalyzed reaction of the in situ generated ortho-bromoacetophenone with the aryl iodide.
- This article is part of the themed collections: Catalysis & biocatalysis in OBC and Direct C-H Functionalization in Method Development and Late Stage Functionalization


 Please wait while we load your content...
Please wait while we load your content...