An ALP-activatable and mitochondria-targeted probe for prostate cancer-specific bimodal imaging and aggregation-enhanced photothermal therapy†
Abstract
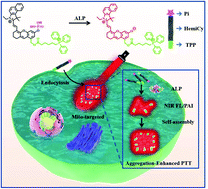
Tumor-derived alkaline phosphatase (ALP) is over-expressed in metastatic prostate cancer. The development of selective probes for ALP detection is therefore critical for early diagnosis and therapy of metastatic prostate cancer. Herein, we develop a mitochondria-targeted near-infrared activatable fluorescent/photoacoustic (NIR FL/PA) probe for the selective detection of prostate cancer-derived ALP and aggregation-enhanced photothermal therapy. Upon dephosphorylation, the probes are activated and they provide a red-shifted strong absorption and emission in the NIR window and thus enable NIR FL and PA imaging of ALP activity in tumor tissues. Particularly, the activated probes self-assemble in situ into a supramolecular network structure which induces cell apoptosis and significantly enhances the photothermal therapy efficacy.


 Please wait while we load your content...
Please wait while we load your content...