Synthesis, structural characterization and computational study of NLO-responsive chromophores and second-order coefficients of thermally crosslinked polymers
Abstract
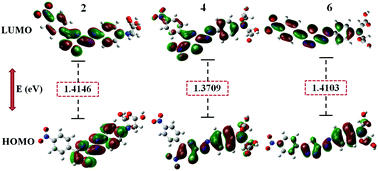
This paper emphasizes the preparation of nonlinear optical (NLO) responsive chromophores and their corresponding polymers. Initially, the carboxyl acid group-based precursors of the chromophores containing strong acceptors such as nitro-substituted thiazole and thiadiazole groups were prepared and subjected to condensation reactions with a monomer to yield a series of polymers. Prior to the synthesis of the polymers, a thermally crosslinkable monomer, 2,6-di-(octy-1-nyl)-p-phenylenediamine, was synthesized by a Sonogashira coupling reaction. All the synthesized precursors, the monomer and their corresponding polymers were characterized using FTIR, 1H NMR, and CHN analyses and UV-Vis spectrophotometry. Simultaneously, the molecular structures of the precursors were designed and optimized using the Gaussian 16 (Revision A.03) program. The dipole moments, energy band gaps between the HOMO and LUMO, polarizabilities and first order hyperpolarizabilities of the precursors were calculated theoretically using the DFT approach; the values were in the ranges of 9.4321–15.3354 D, 1.3709–1.4146 eV, 53.40–64.80 × 10−24 esu and 3703.35–5254.90 × 10−32 esu, respectively. The inherent viscosities (η inh) of the polymers were measured by an Ubbelohde viscometer and were in the range of 0.2453–0.2860 dl g−1. The thermal behavior of the polymers was investigated using DSC and TGA. The glass transition temperatures of the polymers were in the range of 190–218 °C. The thicknesses and refractive indices of the thin films were determined by ellipsometry, and the values were in the range of 0.100–0.127 μm and 1.211–1.426, respectively. The molecular orientations in the thin polymer films were induced by corona poling and were ascertained using both a UV-Vis spectrophotometer and an atomic force microscope. The second harmonic generations (SHGs) of the poled polymers were determined using an Nd-YAG laser. The SHG coefficients (d33) ranged between 98.30 and 106.04 pm V−1 at 532 nm. Among the synthesized polymers, the polymer containing dinitro-substituted thiazole demonstrated an excellent SHG coefficient as high as 106.04 pm V−1. Furthermore, none of the polymers showed SHG decay below 100 °C, and they retained 95% of their SHG values even up to 600 h. Based on its enhanced NLO efficiency and long temporal stability, the polymer containing dinitro-substituted thiazole can be a particularly promising candidate for photonic devices.


 Please wait while we load your content...
Please wait while we load your content...