Development of colloidal rare-earth arsenites as arsenic trioxide nanoparticle prodrugs (ATONP) for chemotherapy on a patient-derived xenograft model of colorectal cancer
Abstract
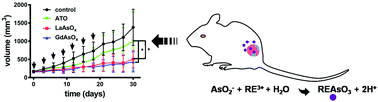
Colloidal gadolinium arsenite has been developed as an arsenic trioxide nanoparticle prodrug (ATONP) by our group in the past five years, and it has illustrated to be highly promising for cancer therapy. Here, we systemically synthesized arsenites of all the rare-earth elements, characterized their element-dependant physicochemical properties (composition, morphology, structure, colloidal charge and acidity), investigated their ATO releasing behavior as well as cytotoxicity, and applied them for the chemotherapy of a patient-derived xenograft model of colorectal cancer. Arsenite colloids of all rare earth elements except those of cerium (Ce) have similar structure, surface charge and stoichiometry ratio between the rare-earth element and arsenic (1 : 0.9–1.2), whereas two dominant morphologies were found. By acting as the prodrugs of arsenic trioxide, they showed similar ATO releasing kinetics against plasma and close cytotoxicity towards the SMMC-7721 liver cancer cell line. Two representative colloidal prodrugs (lanthanide and gadolinium arsenite colloids) were applied in the chemotherapy of a patient-derived colorectal cancer xenograft model, and both ATO prodrugs had a significantly better therapeutic effect than ATO. In addition, they were highly tolerable and showed no significant toxicity. On the other hand, since several rare earth ions act as probe sensitizers (Eu and Tb for luminescence and Gd for MRI) our results may supply a series of functional agents for image-guided chemotherapy.


 Please wait while we load your content...
Please wait while we load your content...