An activation-free route to porous magnetic carbon adsorbents for the removal of phenolic compounds†
Abstract
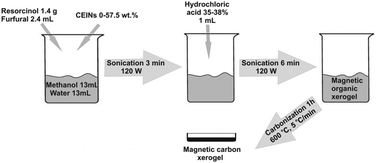
Magnetic composite carbon xerogels were synthesized via acidic gelation of resorcinol and furfural in the presence of carbon-encapsulated iron nanoparticles with subsequent carbonization of the as-obtained magnetic organic xerogel. The carbonization of organic gels resulted in magnetic composite carbon xerogels with highly developed porosity. The presence of carbon-encapsulated iron nanoparticles guaranteed the mobility of the composite xerogels in external magnetic fields and facilitated their separation. The porosity of the synthesized composite xerogels was developed during carbonization and further chemical or physical activation was not required. The adsorption performance of the composite materials obtained was tested in the removal of phenol, 2-chlorophenol and 4-chlorophenol from aqueous solutions. The maximum adsorption capacities were found to be 95, 130 and 138 mg g−1 for phenol, 2-chlorophenol and 4-chlorophenol, respectively. The adsorption kinetics studies showed that the adsorption equilibrium is reached with a contact time of 180 min for phenol and 120 min for its chlorinated derivatives, respectively. The materials presented can be used several times after a simple regeneration procedure. It was also demonstrated that the developed methodology yields magnetic composite adsorbents with high corrosion resistance.


 Please wait while we load your content...
Please wait while we load your content...