The aromaticity of the phenyl ring imparts thermal stability to a supramolecular hydrogel obtained from low molecular mass compound†
Abstract
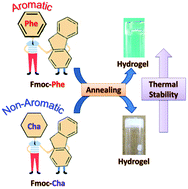
Supramolecular hydrogels obtained from fluorenylmethoxycarbonyl (Fmoc)-functionalized phenylalanine and short peptides containing phenylalanine find potential applications in the biomedical field. Although the significance of the phenyl moiety in hydrogelation of these gelators has been studied, the role of the aromaticity of the phenyl ring is less explored. Therefore, the aim of the present study is to understand the contribution of the aromaticity of the phenyl ring in supramolecular hydrogelation by comparing the hydrogelation between Fmoc-phenylalanine (Fmoc-Phe) and its non-aromatic phenylalanine analog Fmoc-cyclohexylalanine (Fmoc-Cha). The conventional vial inversion test indicated that not only Fmoc-Phe but also Fmoc-Cha equally formed a stable hydrogel when it was subjected to an annealing process, which suggests that the hydrophobicity of the phenyl ring contributes to the formation of the hydrogel rather than the aromaticity. Interestingly, the Fmoc-Phe hydrogel elicits a higher Tgel (∼65 °C) compared to Fmoc-Cha (Tgel = ∼50 °C), which indicates that the aromaticity of the phenyl ring imparted thermal stability to the hydrogel through its additional intermolecular π–π interactions. While the previous report concluded Fmoc-Cha as a non-gelator based on the pH-switch approach, the current result together with the former report thus suggests that the significance of the aromaticity of the phenyl ring in supramolecular hydrogelation by LMM compounds may depend on the method of inducing hydrogelation.


 Please wait while we load your content...
Please wait while we load your content...