Nitrogen and sulfur dual-doped graphene as an efficient metal-free electrocatalyst for the oxygen reduction reaction in microbial fuel cells
Abstract
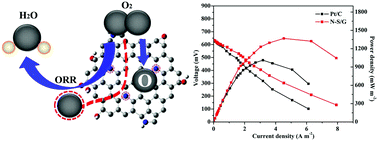
Developing highly efficient non-precious metal electrocatalysts for the oxygen reduction reaction (ORR) is of great importance to obtain high-performance microbial fuel cells (MFCs). In the present work, nitrogen and sulfur dual-doped graphene (N/S-G) was prepared and used as a cathode catalyst for MFCs. The electrocatalytic activity of as-prepared catalysts towards the ORR was evaluated by cyclic voltammetry (CV) and linear sweep voltammetry (LSV). The ORR activity of N/S-G was compared to that of commercial Pt/C in a neutral medium, and this material showed an electron transfer number of 3.75 ± 0.15, suggesting that ORR catalysis proceeded through a favorable four-electron pathway. More importantly, the MFC with N/S-G as a cathode catalyst exhibited a maximum power density of 1368 mW m−2, relatively higher than that obtained with Pt/C (1011 mW m−2). These results demonstrated that N/S-G could be a good candidate as an electrocatalyst for MFCs.


 Please wait while we load your content...
Please wait while we load your content...