Single particle ICP-MS and GC-MS provide a new insight into the formation mechanisms during the green synthesis of AgNPs†
Abstract
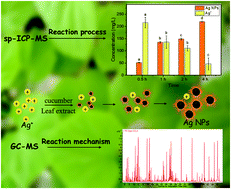
Green synthesis of metallic nanoparticles (NPs) using plant extracts has received considerable attention due to its environmentally and economically friendly nature. Various metabolites in plants such as amino acids, organic acids, sugars and phenolic compounds have been speculated to be responsible for the synthesis of metallic NPs in previous studies. However, to date, there has been a lack of direct evidence linking specific metabolites to the reduction of metal ions to form metallic NPs. Here, AgNPs are synthesized using cucumber leaf extract and characterized by UV-visible spectroscopy, dynamic light scattering (DLS) and transmission electron spectroscopy (TEM). Single particle inductively coupled plasma mass spectrometry (sp-ICP-MS) was used to investigate the size of newly synthesized NPs as well as the kinetics of particle formation. Gas chromatography–mass spectrometry (GC-MS) based metabolomics identified and quantified 245 metabolites in cucumber leaf extracts. By comparing the concentrations of metabolites before and after the reaction, the metabolites responsible for the synthesis were screened out. Reducing sugars (cellobiose, ribulose-5-phosphate, melibiose, 2-deoxy-D-glucose, tagatose, fructose, ribose, 3,6-anhydro-D-galactose) were markedly decreased after the reaction, indicating that reducing sugars are involved in the biosynthesis process and possibly function as reducing agents. The key thermodynamic data of the reaction between Ag+ and reducing sugars were obtained by using isothermal titration calorimetry (ITC), which further confirmed the interaction between Ag+ and metabolites. This study provides a deep insight into the reaction process and mechanism of green synthesized AgNPs.


 Please wait while we load your content...
Please wait while we load your content...