What is the effect of Sn and Mo oxides on gold catalysts for selective oxidation of benzyl alcohol?†
Abstract
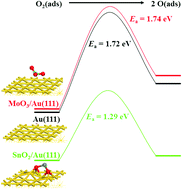
A foam-like mesoporous silica (MCF)-supported gold (Au) nanoparticle (NP) catalyst was prepared by a facile one-pot synthesis strategy. SnOx and MoOx were introduced as promoters by a wet impregnation method to synthesize promoted catalysts Au–SnOx/MCF and Au–MoOx/MCF, respectively. The evaluation of the prepared catalysts in benzyl alcohol oxidation shows that introduction of a low amount of SnOx (0.2 wt%) leads to a higher catalytic activity, while a relatively high amount of SnOx (1.0 wt%) exhibits a lower catalytic activity than the unpromoted Au NP catalyst. Both characterization results and density functional theory (DFT) calculations suggest that the most important effect of SnOx species on gold catalysts is the strong interaction between them which leads to electron transfer and lattice distortion of Au NPs, providing more low-coordinated Au active sites. This structural modification enhanced the chemisorption of reactants and lowered the energy barrier for the dissociation of O2 molecules on the surface of gold catalysts, improving the catalytic performance for benzyl alcohol oxidation. However, a weaker interaction was detected between MoOx and Au NPs on catalysts Au–MoOx/MCF and no lattice modification was observed; thus no improvement in the catalytic activity for benzyl alcohol oxidation occurred on catalysts Au–MoOx/MCF.


 Please wait while we load your content...
Please wait while we load your content...