Kinetic, isotherm and mechanism studies of organic dye adsorption on poly(4,4′-oxybisbenzenamine) and copolymer of poly(4,4′-oxybisbenzenamine-pyrrole) macro-nanoparticles synthesized by multifunctional carbon dots†
Abstract
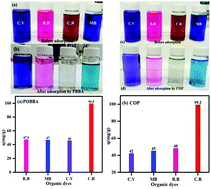
Poly(4,4′-oxybisbenzenamine) (POBBA) and poly(4,4′-oxybisbenzenamine-pyrrole) (COP) with mixed macro–nano sizes were synthesized by a facile one-step reaction using multifunctional carbon dots as the initiator. The two polymers were proven as effective adsorbents for four popular organic dyes in solution: Congo red, crystal violet, methylene blue, and rhodamine B. The adsorption between the adsorbate and the adsorbent was probed by Fourier transform infrared spectroscopy (FTIR). The adsorption mechanism was governed by the pseudo-second-order model, whereas the Freundlich isotherm fitted the adsorption data well for both adsorbent materials.


 Please wait while we load your content...
Please wait while we load your content...