Non-invasive sensing of transepithelial barrier function and tissue differentiation in organs-on-chips using impedance spectroscopy†
Abstract
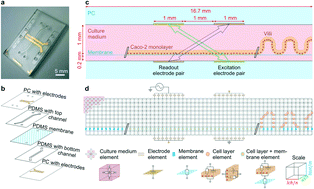
Here, we describe methods for combining impedance spectroscopy measurements with electrical simulation to reveal transepithelial barrier function and tissue structure of human intestinal epithelium cultured inside an organ-on-chip microfluidic culture device. When performing impedance spectroscopy measurements, electrical simulation enabled normalization of cell layer resistance of epithelium cultured statically in a gut-on-a-chip, which enabled determination of transepithelial electrical resistance (TEER) values that can be compared across device platforms. During culture under dynamic flow, the formation of intestinal villi was accompanied by characteristic changes in impedance spectra both measured experimentally and verified with simulation, and we demonstrate that changes in cell layer capacitance may serve as measures of villi differentiation. This method for combining impedance spectroscopy with simulation can be adapted to better monitor cell layer characteristics within any organ-on-chip in vitro and to enable direct quantitative TEER comparisons between organ-on-chip platforms which should help to advance research on organ function.
- This article is part of the themed collections: A Special Collection in honour of Albert van den Berg and Organ-, body- and disease-on-a-chip systems


 Please wait while we load your content...
Please wait while we load your content...