Analytical methodology for the simultaneous determination of NMG-Sb(v), iSb(v), and iSb(iii) species by anion exchange liquid chromatography in Glucantime® and its biological application in Wistar rat urine†
Abstract
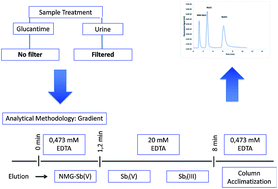
Several methodologies have been studied to determine the species of antimony (Sb) present in Glucantime®. However, these techniques have certain disadvantages, such as a low resolution between N-methylglucamine (NMG-Sb(V)) and inorganic pentavalent Sb (iSb(V)). Furthermore, there is a problem regarding the stability of inorganic trivalent Sb (iSb(III)) during sample treatment. The main aim of this work was to develop an analytical methodology for the separation of the Sb species of NMG-Sb(V), iSb(V), and iSb(III) in Glucantime®. To achieve this, a methodology was developed based on anion exchange high performance liquid chromatography hydride generation atomic fluorescence (HPLC-HG-AFS) in gradient elution mode with different concentrations of Na2H2EDTA as the mobile phase. The results show that 0.473 mM Na2H2EDTA should be used for 1.20 min as the first mobile phase for the separation of NMG-Sb(V), which should then be changed to 20 mM for the elution of the inorganic species. The results also reveal that the iSb(III) content may be underestimated when sample filtration is used prior to the separation step. Instead of filtration, this study demonstrates that adjustment to pH 9 and dilution with deionized water are a better strategy to determine the iSb(III) content. This method proves to be reproducible for application in urine samples from rats injected intraperitoneally with NMG-Sb(V).
- This article is part of the themed collection: Analytical Atomic Spectrometry in South America


 Please wait while we load your content...
Please wait while we load your content...