Synthesis of high-molecular weight isosorbide-based polycarbonates through efficient activation of endo-hydroxyl groups by an ionic liquid†
Abstract
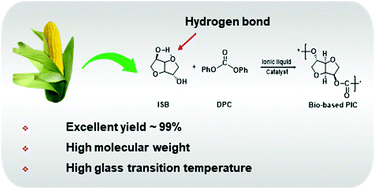
The utilization of renewable resources and carbon dioxide (CO2) has attracted extensive attention due to global warming and continuously declining petroleum-based resources. Herein, a novel synthetic approach of bio-based polycarbonates using isosorbide (ISB) and diphenyl carbonate (DPC), derived from the renewable resources glucose and CO2, respectively, was proposed, where an ionic liquid (IL) was used as a promising green catalyst; to overcome the poor activity of the endo-hydroxyl group (endo-OH) of ISB and achieve high-molecular weight polycarbonates, a new strategy was revealed that involved the activation of the endo-OH group via a series of efficient tetrabutylphosphonium-based IL catalysts. By designing the structures of cations and anions, most of the endo-OH groups have been activated and incorporated into the polymer chain, and the ratio of endo-OH/exo-OH at the end group of the polymer chain has been decreased to 0.25, which, to the best of our knowledge, is the lowest value reported to date. Among the prepared ILs, tetrabutylphosphonium acetate in a trace amount (1.5 × 10−6 mol) exhibited highest catalytic performance, and poly(isosorbide carbonates) (PICs) with the molecular weight of up to 66 900 g mol−1, Tg of 169 °C and better thermal stability than conventional polycarbonates were achieved. Mass spectrometry and density functional theory calculations provided a deeper understanding of the underlying cation–anion synergistic catalytic mechanism. Furthermore, copolymerization of ISB, diols and DPC was performed to modify the flexibility of PIC, resulting in copolycarbonates with the high molecular weight of 110 000 using 1,10-decanediol as a copolymer monomer.


 Please wait while we load your content...
Please wait while we load your content...