Efficient removal of Pb2+ by Tb-MOFs: identifying the adsorption mechanism through experimental and theoretical investigations†
Abstract
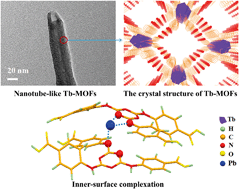
Nanotube-like Tb-based metal–organic frameworks (Tb-MOFs) were synthesized via the assembly of carboxylate-based ligands and Tb3+ under solvothermal conditions. The porous Tb-MOFs with high stability and considerable active functional groups make them ideal adsorbents in environment remediation. The factors influencing the adsorption property of Tb-MOFs toward Pb2+ ions were studied, comprising pH, ionic strength, adsorbent content, initial Pb2+ concentration and contact time. The Tb-MOFs exhibited excellent adsorption property with a maximum removal capacity of 547 mg g−1 and could maintain a high adsorption performance even after five cycles. Results from batch adsorption experiments and X-ray photoelectron spectroscopy (XPS) analysis imply that the formation of the inner-sphere complex (C–/![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N⋯Pb) between the nitrogenous groups of Tb-MOFs and Pb2+ is the primary adsorption mechanism. Furthermore, density functional theory (DFT) calculations confirm that the most favorable adsorption configuration varies with the reaction conditions and deprotonated functional groups tend to bond with Pb2+ at high pH. These results suggest that Tb-MOFs could be used as a promising adsorbent for the removal of Pb2+ ions from an aqueous environment.
N⋯Pb) between the nitrogenous groups of Tb-MOFs and Pb2+ is the primary adsorption mechanism. Furthermore, density functional theory (DFT) calculations confirm that the most favorable adsorption configuration varies with the reaction conditions and deprotonated functional groups tend to bond with Pb2+ at high pH. These results suggest that Tb-MOFs could be used as a promising adsorbent for the removal of Pb2+ ions from an aqueous environment.


 Please wait while we load your content...
Please wait while we load your content...