Valence tautomerism and spin crossover in pyridinophane–cobalt–dioxolene complexes: an experimental and computational study†
Abstract
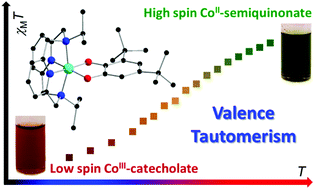
Compounds [Co(L-N4R2)(dbdiox)](BPh4) (L-N4R2 = N,N′-di-alkyl-2,11-diaza[3.3]-(2,6)pyridinophane, R = iPr (1a), Et (2a); dbdiox = 3,5-di-tert-butyldioxolene) and [M(L-N4iPr2)(dbdiox)](BPh4) (M = Mn (3a), Fe (4a)) have been synthesized and investigated with a view to possible valence tautomeric (VT) or spin crossover (SCO) interconversions. Single crystal X-ray diffraction data for all compounds at 100 or 130 K indicate trivalent metal cations and di-tert-butylcatecholate (dbcat2−) dioxolene ligands. Variable temperature magnetic susceptibility data for all species between 2 and 340 K are consistent with these redox states, with low spin configurations for the cobalt(III) ions and high spin for the manganese(III) and iron(III) ions. Above 340 K, compound 1a exhibits an increase in magnetic susceptibility, suggesting the onset of a VT interconversion from low spin Co(III)-dbcat to high spin Co(II)-dbsq (dbsq− = di-tert-butylsemiquinonate) that is incomplete up to 400 K. In solution, variable temperature electronic absorption spectra and Evans NMR method magnetic susceptibility data indicate reversible VT interconversions for 1a in several solvents, with the transition temperature varying with solvent. Variable temperature electronic absorption spectra are temperature-invariant for 3a and 4a, while spectra for 2a in 1,2-dichloroethane suggest the onset of a VT transition at the highest temperatures measured. Density functional theory calculations on all four compounds and literature analogues provide key insights into the relative energies of the different electromeric forms and the possibilities for VT versus SCO interconversions in this family of compounds.


 Please wait while we load your content...
Please wait while we load your content...