Synthesis and characterization of chromium complexes 2-Me4CpC6H4CH2(R)NHCrCl2 and their catalytic properties in ethylene homo- and co-polymerization†
Abstract
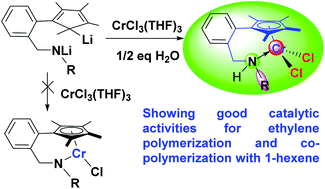
A series of new half-sandwich secondary amine-coordinated dichlorochromium complexes chelated by 2-(tetramethylcyclopentadienyl)benzylamine ligands, 2-Me4CpC6H4CH2(R)NHCrCl2 [R = iPr (1), Cy (2), Ph (3), 4-MePh (4), 2,6-Me2Ph (5), 2,6-Et2Ph (6)], have been synthesized from the reactions of CrCl3(THF)3 with the dilithium salts of the corresponding ligands in THF, followed by the addition of 1/2 eq. of H2O to the reaction mixtures. The isolated yields of the chromium complexes were found to increase with the increase in the amount of H2O introduced and reach the highest values (66–76%) when 1/2 eq. of H2O is added. Attempts to isolate the 2-(tetramethylcyclopentadienyl)benzylamidochromium complexes, 2-Me4CpC6H4CH2(R)NCrCl, were not successful. The new dichlorochromium complexes were characterized by IR, 1H NMR, EPR, and UV/Vis spectroscopy and elemental analyses, and the molecular structures of complexes 1, 5 and 6 were determined by X-ray crystallography. The X-ray crystallographic analysis reveals that these chromium complexes possess a three-legged piano-stool geometry with the amine N atom in a mitered six-membered chelating ring and the two chloride atoms as the legs. Upon activation with AlR3 and Ph3CB(C6F5)4, complexes 1–6 exhibit reasonable catalytic activity for ethylene polymerization and copolymerization with 1-hexene, producing polyethylenes with moderate to high molecular weights and poly(ethylene-co-1-hexene)s with moderate comonomer incorporation which are typical linear low-density polyethylenes (LLDPE). Complex 4 was found to show higher catalytic activity for ethylene homo- and co-polymerization than other complexes under similar conditions, while complex 3 produced poly(ethylene-co-1-hexene)s with the highest comonomer incorporation.


 Please wait while we load your content...
Please wait while we load your content...