Variable oxidation state sulfur-bridged bithiazole ligands tune the electronic properties of ruthenium(ii) and copper(i) complexes†
Abstract
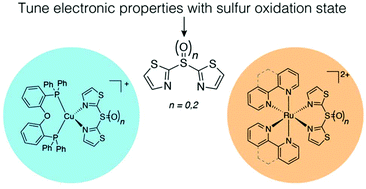
The synthesis of homoleptic and heteroleptic ruthenium(II) and copper(I) complexes containing sulfur-bridged bithiazole ligands of varying oxidation states are reported. The complexes have been characterized using 1D and 2D NMR spectroscopy, X-ray single crystal diffraction, electrochemistry, UV-vis absorbance and fluorescence spectroscopy. The stability, photophysical and electrochemical properties were found to be dependent on the oxidation state of the non-coordinating sulfur. The ruthenium and copper species were found to be non-emissive in solution at room temperature, though all displayed weak emission when doped in a PMMA matrix, which increased in intensity on cooling to 77 K. The electrochemical HOMO–LUMO gap was found to be dependent on the oxidation state of the sulfur in the bridging ligand in all complexes, illustrating an additional handle for tuning electrochemical properties.


 Please wait while we load your content...
Please wait while we load your content...