Study on micro-nanocrystalline structure control and performance of ZnWO4 photocatalysts
Abstract
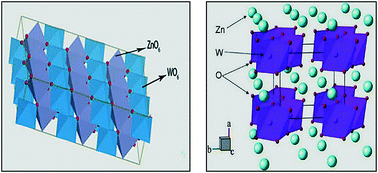
ZnWO4 micro/nanocrystals with different sizes and well-developed crystals were synthesized by the molten salt method at low temperature. The crystal structure and growth morphology of the ZnWO4 crystals were characterized and the crystalline and band structure of ZnWO4 was simulated using Materials Studio software to reveal the relationship between crystal structure, surface morphology and properties. The results show that the ZnWO4 single crystal can be obtained at 500 °C for 12 h. The photocatalytic activity of the nanocrystalline ZnWO4 crystals prepared at different calcining times is decreased in the order 12 h > 24 h > 6 h > 0 h and the fluorescence emission intensity of the ZnWO4 crystals prepared at 500 °C for 24 h exhibited strong photoluminescence properties. The photocatalytic and photoluminescence properties of the ZnWO4 crystals were closely related to the crystal structure, micromorphology and crystal size. Based on the theoretical calculation of the energy band structure, the photoexcited electrons transmitted from the O 2p orbitals at the top of the VB to the empty W 5d orbitals at the bottom of the CB and the larger photoelectric transmission rate can inhibit the recombination of electron–hole pairs and promote the photocatalytic reaction. In contrast, the binding and complexation of carriers contributed to the manifestation of photoluminescence performance.


 Please wait while we load your content...
Please wait while we load your content...