Persistent homology analysis of osmolyte molecular aggregation and their hydrogen-bonding networks
Abstract
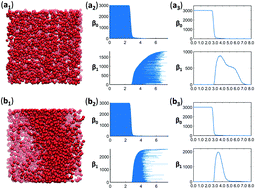
Dramatically different properties have been observed for two types of osmolytes, i.e., trimethylamine N-oxide (TMAO) and urea, in a protein folding process. Great progress has been made in revealing the potential underlying mechanism of these two osmolyte systems. However, many problems still remain unsolved. In this paper, we propose to use the persistent homology to systematically study the osmolytes’ molecular aggregation and their hydrogen-bonding network from a global topological perspective. It has been found that, for the first time, TMAO and urea show two extremely different topological behaviors, i.e., an extensive network and local clusters, respectively. In general, TMAO forms highly consistent large loop or circle structures in high concentrations. In contrast, urea is more tightly aggregated locally. Moreover, the resulting hydrogen-bonding networks also demonstrate distinguishable features. With a concentration increase, TMAO hydrogen-bonding networks vary greatly in their total number of loop structures and large-sized loop structures consistently increase. In contrast, urea hydrogen-bonding networks remain relatively stable with slight reduction of the total loop number. Moreover, the persistent entropy (PE) is, for the first time, used in characterization of the topological information of the aggregation and hydrogen-bonding networks. The average PE systematically increases with the concentration for both TMAO and urea, and decreases in their hydrogen-bonding networks. But their PE variances have totally different behaviors. Finally, topological features of the hydrogen-bonding networks are found to be highly consistent with those from the ion aggregation systems, indicating that our topological invariants can characterize intrinsic features of the “structure making” and “structure breaking” systems.


 Please wait while we load your content...
Please wait while we load your content...