Unveiling the thermolysis natures of ZIF-8 and ZIF-67 by employing in situ structural characterization studies†
Abstract
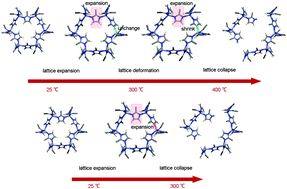
The thermolysis routes of two isostructural metal–organic framework compounds (Zn-based ZIF-8 and Co-based ZIF-67) are investigated based on temperature-dependent and time-dependent in situ Fourier transform infrared (FTIR) spectroscopy and in situ X-ray diffraction data, as well as thermogravimetric-differential scanning calorimetry (TG-DSC) analyses and density functional theory (DFT) calculations. These data highlight thermolysis effects on different vibrations and dissociations within specific atomic moieties. The coordination differences between Zn–N and Co–N lead to the distinct thermolysis routes of ZIF-8 and ZIF-67. ZIF-8 is easily deformed during heating while decomposes at a higher temperature due to the saturated Zn–N coordination. ZIF-67, however, does not deform during heating due to the stronger Co–N bonds, but easily reacts with oxygen due to the unsaturated Co–N bonds. Our results demonstrate that in situ FTIR paired with in situ XRD is a powerful technique for MOF thermolysis investigation, and we suggest that the thermolysis mechanisms of MOFs may be unveiled by investigating a series of MOFs having different coordination types using in situ characterisation methods.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...