Does induced current density explain the C–H and C–F Perlin effects?†
Abstract
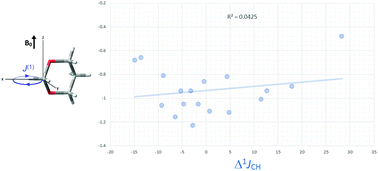
Spin–spin coupling constant (SSCC) data may be useful in providing information on the stereochemistry and intramolecular interactions in molecules. One-bond C–H and C–F SSCCs (1JCH and 1JCF) are amongst the most important NMR parameters used to study the structure of alicyclic six-membered rings, because the Perlin effect, defined as 1JCHeq > 1JCHax, and the fluorine Perlin-like effect, defined as |1JCFeq| > |1JCFax|, are in wide currency to probe the conformations of these compounds. The origin of these effects has been usually attributed either to dipolar interactions or hyperconjugation, while the induced current density (ICD) has been recently correlated to the magnetic shielding tensors in some six-membered ring compounds, and then used to explain the Perlin effect. Accordingly, this work reports an analysis of the ICD as a descriptor of 1JCH and 1JCF in a series of six-membered rings to find out the role of the ICD in the conventional and fluorine Perlin effect. The atoms in molecules (AIM) magnetic responses obtained from density functional theory (DFT) calculations for the studied compounds did not show any relationship of the first-order electronic current density (ΔJ(1)) with the calculated Δ1JCHax-eq and Δ1JCFax-eq values. Consequently, the title effects cannot be precisely explained by ICD. Nevertheless, an interesting relationship between ΔJ(1) and Δdelocalization involving σCH* orbitals is observed.


 Please wait while we load your content...
Please wait while we load your content...