Ligand stabilization of manganocene dianions – in defiance of the 18-electron rule†
Abstract
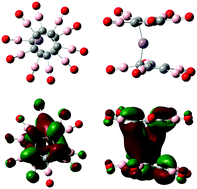
Manganocene [Mn(C5H5)2], a 17-electron system, is expected to have a high electron affinity, as addition of an extra electron would make it a closed-shell 18-electron system. Surprisingly, it has a very low electron affinity of only 0.28 eV. Combined with its high ionization potential of around 7.0 eV, manganocene, therefore, should not be eager to either donate or accept an electron. We show that this property can be fundamentally altered with the proper choice of ligands, even though the total electron count remains the same. For example, the electron affinities of manganocene-derivatives Mn[C5(CN)5]2 and Mn[C5(BO)5]2, created by replacing H with CN or BO, are found to be 4.78 eV and 4.85 eV, respectively, making these species superhalogens. The power of the ligands is further demonstrated by studying the stability of their di-anions. Note that [Mn(C5X5)2]2− (X = H, CN, BO) di-anions, with 19-electrons, have one electron more than necessary to satisfy the 18-electron rule for stability. This factor, combined with the unavoidable repulsion between the two extra electrons, should destabilize [Mn(C5X5)2]2−. While that is the case for [Mn(C5H5)2]2−, we show that both Mn[C5(CN)5]22− and Mn[C5(BO)5]22− are stable against auto-detachment of the second electron by 0.7 eV and 0.38 eV, respectively. These results, based on first-principles calculations, demonstrate that ligand-manipulation can be used as an effective strategy to design and synthesize new materials with novel and tailored properties.


 Please wait while we load your content...
Please wait while we load your content...