The effect of protons on the Mg2+ migration in an α-V2O5 cathode for magnesium batteries: a first-principles investigation†
Abstract
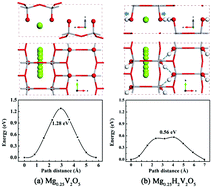
The scarce inventory of cathode materials with reasonable diffusion of Mg ions is the main obstacle in the development of rechargeable magnesium batteries. In this regard, vanadium pentoxide (V2O5) has been reported to be a candidate cathode material for Mg batteries. In this study, via first-principles calculations, we showed that the Mg-ion diffusion energy barrier in α-V2O5 could be substantially decreased through hydrogenation. It is found that the Mg-ion migration energy barrier in HxV2O5 is gradually decreased with an increase in H concentration. When the H concentration x reaches 2, the migration barrier is decreased to 0.56 eV from that in α-V2O5 without hydrogenation (1.28 eV). This indicates that the Mg diffusion kinetics can be substantially improved through hydrogenation, and the resultant energy barrier makes Mg diffusion acceptable even at room temperature. The mechanism of the H-enhanced Mg-diffusion has also been studied, and it has been found that H atoms not only can expand the Mg-diffusion pathway, but also have a screening effect on the interactions between Mg ions and the α-V2O5 lattice.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...