Entangling non planar molecules via inversion doublet transition with negligible spontaneous emission†
Abstract
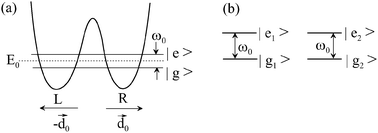
We analyze theoretically the entanglement between two non-planar and light identical molecules (e.g., pyramidal NH3) that present inversion doubling due to the internal spatial inversion of their nuclear conformations by tunneling. The peculiarity of this system lies in the simplicity of this type of molecular system in which two near levels can be connected by an allowed electric dipole transition with considerable value of the dipole moment transition and negligible spontaneous emission because the transition is in the microwave or far-infrared range. These properties give place to entanglement states oscillating by free evolution with frequency determined by the dipole–dipole interaction and negligible spontaneous decay, which allows consideration of an efficient quantum Zeno effect by frequent measurements of one of the entangled states. If the molecules are initially both in the upper (or lower) eigenstate, the system evolves under an external radiation field, which can induce oscillations of the generated entangled states, with frequency of the order of the Rabi frequency of the field. For a certain detuning, a symmetric entangled state, which is an eigenstate of the collective system, can be populated, and given its negligible spontaneous emission, could be maintained for a time limited only by external decoherence processes, which could be minimized. Although the data used are those of the NH3 molecule, other molecules could present the same advantageous features.


 Please wait while we load your content...
Please wait while we load your content...