A microporous zinc–organic framework with Lewis basic pyridyl sites for highly selective C2H2/CH4 and C2H2/CO2 gas separation†
Abstract
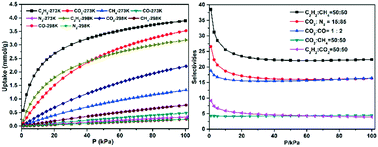
A new Zn-based metal–organic framework with 8-connected hex topology constructed from bipyridine and carboxylate ligands, namely [Zn(cpna)3(tmbpy)]n·4DMF (1) (H2cpna = 6-(4-carboxyphenyl)nicotinic acid; tmbpy = 3,3′-dimethyl-4,4′-bipyridine), has been synthesized under solvothermal conditions. The Zn-MOF was structurally characterized and retained a good crystal structure after activation (1a), an indication of permanent porosity. 1a exhibits a BET surface of 770 m2 g−1 and the maximum aperture of 8.60 Å. Single component adsorption isotherm measurements of C2H2, CO2, CH4, CO and N2 on 1a were performed both at 273 K and 298 K, and the selectivities for C2H2/CH4, C2H2/CO2, CO2/CO, CO2/CH4 and CO2/N2 were estimated on the basis of the ideal adsorbed solution theory (IAST). 1a shows the uptake of C2H2 (3.89 mmol g−1) and CO2 (3.52 mmol g−1) at 273 K and 100 kPa, respectively. Due to the Lewis basic pyridyl sites inside the channel of the framework, 1a showed good adsorption selectivities for C2H2/CH4(50 : 50) and C2H2/CO2(50 : 50) at 298 K (100 kPa).


 Please wait while we load your content...
Please wait while we load your content...