Theobromine cocrystals with monohydroxybenzoic acids – synthesis, X-ray structural analysis, solubility and thermal properties†
Abstract
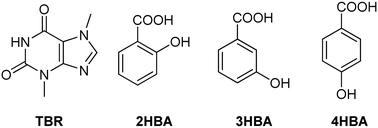
Theobromine, an organic compound from the purine alkaloid group, is much less soluble in polar solvents than its analogues, i.e. caffeine and theophylline, that is why it has been used as an active pharmaceutical ingredient (API) model in cocrystal preparation. A series of theobromine (TBR) cocrystallization processes from solutions with such coformers as 2-hydroxybenzoic acid (2HBA), 3-hydroxybenzoic acid (3HBA) and 4-hydroxybenzoic acid (4HBA) were carried out. In addition, neat grinding and liquid-assisted grinding were performed. The obtained cocrystals TBR·2HBA and TBR·3HBA as well as TBR·2(4HBA)·H2O cocrystal monohydrate have been characterized by single crystal X-ray diffraction (SXRD), PXRD, UV-vis and STA (TGA/DSC) analyses. In all cases no proton transfer from the acid molecule to the imidazole nitrogen atom in theobromine was observed. TBR–acid heterosynthons are sustained by N⋯H–O interactions, where proton donors in TBR·2HBA and TBR·3HBA are carboxylic groups, and in TBR·2(4HBA)·H2O the proton donor is the hydroxyl group of the acid molecule. In each cocrystal, TBR–TBR homosynthon R22(8) formation by N–H⋯O hydrogen bonds was observed. Acid–acid dimers are created only in the crystal lattice of TBR·2(4HBA)·H2O. In the obtained cocrystals, similar supramolecular synthons were observed, such as in theophylline and caffeine cocrystals with the same coformers. C–H⋯O and π⋯π forces present in the described structures are responsible for 2D and 3D structure stabilization.
- This article is part of the themed collection: 1st International Conference on Noncovalent Interactions


 Please wait while we load your content...
Please wait while we load your content...