Charge-transfer complexes of sulfur-rich acceptors derived from birhodanines†
Abstract
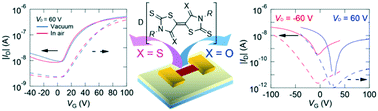
Sulfur-rich acceptors, birhodanines, 3,3′-dialkyl-5,5′-bithiazolidinylidene-2,2′-dione-4,4′-dithiones (OS-R, R = Et and Pr) and 3,3′-dialkyl-5,5′-bithiazolidinylidene-4,4′-dione-2,2′-dithiones (SO-R, R = Et), as well as the sulfur analogues, 3,3′-dialkyl-5,5′-bithiazolidinylidene-2,4,2′,4′-tetrathiones (SS-R, R = Et, Pr), form 1 : 1 composition charge-transfer complexes with donors such as pyrene, perylene, and coronene. These complexes have mixed stacks, and the SS-R complexes show n-channel transistor properties due to the intercolumnar S⋯S contacts between the acceptors. By contrast, the OS-R and SO-R complexes exhibit basically hole-dominant ambipolar properties due to the absence of S⋯S contacts. Accordingly, the charge transport is governed mostly by the direct interchain interactions instead of the transport along the columns.


 Please wait while we load your content...
Please wait while we load your content...