Electrochemical glucose sensing characteristics of two-dimensional faceted and non-faceted CuO nanoribbons
Abstract
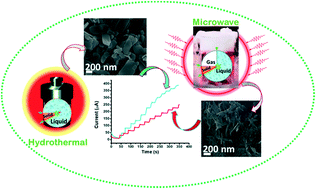
We present faceted and non-faceted crystal cupric oxide (CuO) nanoribbons synthesized by different processes for glucose-sensing applications. The faceted and non-faceted CuO nanoribbons are grown via hydrothermal and microwave heating processes, respectively. Their growth mechanisms are considered as surface energy kinetics supported by a chemical simulation process. Both methods follow a redox precipitation mode for the formation of the nanostructures. Both faceted and non-faceted CuO nanostructures are revealed to exhibit different morphologies, but they have a single crystal structure. In a non-enzymatic glucose sensor, the amperometric oxidation currents of both faceted and non-faceted CuO electrodes increase proportionally as the glucose concentration increases gradually from 0.05 to 3.5 mM. However, the faceted CuO exhibits higher crystallinity with much more surface-active sites as faceted crystal planes compared to the non-faceted ones. Therefore, the glucose sensitivity of a faceted CuO electrode is superior to that of a non-faceted CuO one. The electrochemical glucose detection of the faceted CuO electrode shows a minimum concentration of 58 μM and a specific current sensing of 412 μA mM−1 cm−2, whereas the non-faceted CuO electrode shows low sensitivities of 71 μM and 356 μA mM−1 cm−2.


 Please wait while we load your content...
Please wait while we load your content...