Design of 4-aminobenzoic acid two-component molecular crystals: prediction and experiments†
Abstract
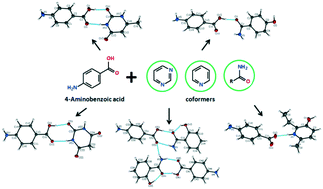
Cocrystal formation of 4-aminobenzoic acid (PABA) has been screened with a variety of pyrimidine, pyridine and benzamide derivatives to understand how hydrogen bond donor/acceptor ratios and the position of a substituting group of the coformers affect cocrystallization. Four new cocrystals with 6-methyluracil (6-MeUr), barbituric acid (BarbAc), 2-hydroxybenzamide (2-OHBZA), 4-hydroxybenzamide (4-OHBZA) and one salt with emoxypine (EMX) have been successfully obtained from solvent evaporation experiments, and their crystal structures have been determined. Analysis of the hydrogen bonding motifs has shown that all the two-component crystals, except for the [PABA + 4-OHBZA] cocrystal, feature acid⋯amide or acid⋯pyridine heterosynthons between the components. The melting temperatures and crystal lattice energy values of the novel PABA multi-component crystals have been described. The temperature dependences of the 6-MeUr and EMX saturation vapour pressure have been studied, and the sublimation thermodynamic functions have been calculated. The thermodynamic functions of cocrystal formation based on PABA have been calculated and analyzed by the diagram method.
- This article is part of the themed collection: The Solid State of Pharmaceuticals


 Please wait while we load your content...
Please wait while we load your content...