Insight into the role of piperazine in the thermodynamics and nucleation kinetics of the triethylenediamine–methyl tertiary butyl ether system†
Abstract
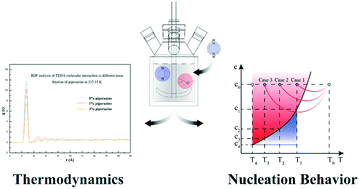
In this work, we studied the effect of piperazine, a major by-product, acting as an additive, on the thermodynamics and nucleation kinetics of the triethylenediamine (TEDA)–methyl tertiary butyl ether (MTBE) crystallization process. It was found that the solubility of TEDA in MTBE increased with increasing amount of piperazine. The interactions of piperazine, TEDA and MTBE were investigated by using molecular dynamics simulation based on Materials Studio. The experimental data of maximum supercooling ΔTmax, a measurement of the metastable zone width (MSZW), were analyzed to describe the role of piperazine in the nucleation kinetics of the TEDA–MTBE system. Modified Sangwal's theory was also employed to understand the nucleation behavior in both pure and impure systems. Improved solubility and narrower MSZWs can be achieved by controlling the amount of piperazine in the TEDA–MTBE system. Based on the thermodynamics and nucleation results, a TEDA product with a large particle size can be obtained by adjusting the supersaturation in a one-step purification process, eliminating the conventional recrystallization step.


 Please wait while we load your content...
Please wait while we load your content...