Amine-induced phase transition from white phosphorus to red/black phosphorus for Li/K-ion storage†
Abstract
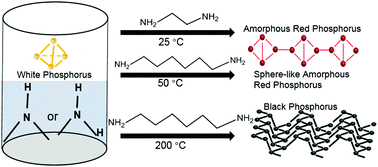
An amine-induced phase transition strategy is developed for converting white phosphorus into red or black phosphorus. Amorphous red phosphorus is prepared through the chemical dissolution of white phosphorus in amines and post-treatment at room temperature. It is demonstrated that the phase transformation mechanism involves an amine-induced nucleophile attack on white phosphorus and the generation of polyphosphorus ions. The amorphous phosphorus exhibits a specific capacity of 955.5 mA h g−1 at 1.5 A g−1 after 1000 cycles, and a capacity retention of 1210.2 mA h g−1 even at 20 A g−1 for Li-ion batteries. This methodology is also applicable to produce red phosphorus micro-spheres and black phosphorus particles by varying the types of organic amines and the reaction temperature.


 Please wait while we load your content...
Please wait while we load your content...