Photo-accelerated “click” reaction between diarylsydnones and ring-strained alkynes for bioorthogonal ligation†
Abstract
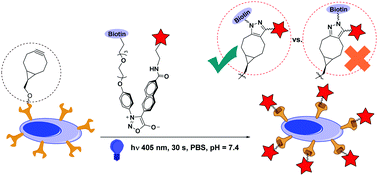
We constructed a library of diarylsydnone (DASyd) candidates in search of a photoclickable reaction toward alkynes, enabling an ultra-accelerated reactivity, while suppressing the background cycloaddition in the dark. The in vitro and in vivo protein labelling experiments revealed that the photo-accelerated DASyd–alkyne cycloaddition exhibits robust selectivity.


 Please wait while we load your content...
Please wait while we load your content...