A facile preparation of functional cycloalkynes via an azide-to-cycloalkyne switching approach†
Abstract
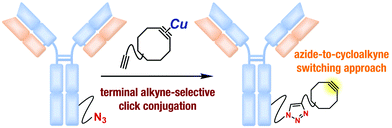
A facile method for preparing various functional cycloalkynes, including proteins incorporated with a cycloalkyne moiety, from the corresponding azides is developed. Treatment of diynes bearing strained and terminal alkyne moieties with a copper salt enabled terminal alkyne-selective click conjugation with azides, whereas a more azidophilic strained alkyne moiety was transiently protected from the click reaction via complexation with copper.


 Please wait while we load your content...
Please wait while we load your content...