Surface sulfonates lock serum albumin into a “hard” corona†
Abstract
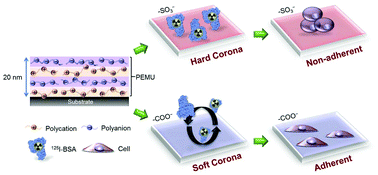
The composition of the layer of proteins adsorbed to macro- or microscopic surfaces of synthetic origin influences the response of living systems to these materials. This adsorbed layer of proteins usually comprises a “soft” coating or corona of labile or exchangeable adsorbed proteins on top of a more tenaciously held “hard” corona in contact with the surface. Here, we link the dependence of cell adhesion on a 20 nm film of polyelectrolyte complex to the “hardness” of the initial corona using albumin, the most prevalent protein in serum. The ease with which albumin can be lost depends on the surface functional group – carboxylate or sulfonate, in particular aromatic sulfonate. Carboxylate permits easier loss of albumin, which presumably allows the subsequent adsorption of proteins such as fibronectin, required for cell adhesion. Sulfonate holds on to albumin more strongly, producing a persistent hard corona likely to remain biocompatible. The mechanism is thought to be related to the higher energy of interaction between sulfonate and amine than between carboxylate and amine, and provides insight on possible reasons why so-called “tissue culture plastic” works so well for in vitro cell culture.


 Please wait while we load your content...
Please wait while we load your content...